 
“Other clinical trials are looking at combining different subtypes of influenza (A and B) to develop a broadly protective vaccine or, in some cases, even a universal flu vaccine, thus avoiding the risk of not having selected the right strain for the next flu season. One key area of interest is combining influenza and COVID-19 vaccines in a single shot. 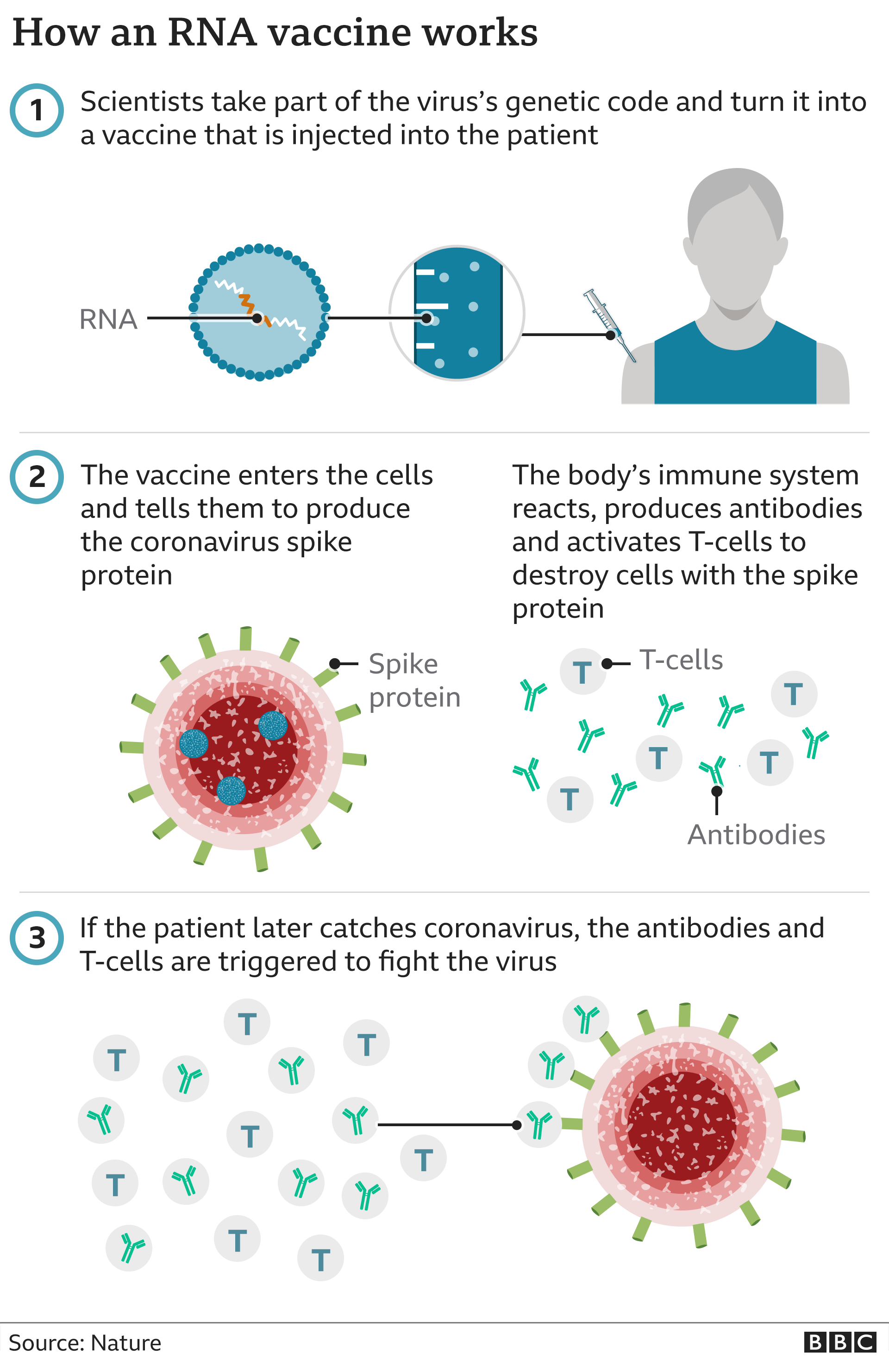
Several companies are now keenly exploring the potential of mRNA. Ultimately, they hope that an mRNA-based method will be faster to develop and manufacture, thus achieving more accurate matching against circulating strains. The story of the COVID-19 collaboration is history, but what of influenza? The work continues – with volunteers recruited for a phase III clinical trial last year. In 2020, the companies agreed to expand their collaboration to the far more pressing threat of COVID-19. The famous Pfizer/BioNTech COVID-19 vaccine actually has its roots in influenza innovation efforts, with the companies agreeing their first partnership in 2018. Recognizing the challenges, some companies were already taking action. “The term in the vaccine industry that is extraordinarily important is ‘clock speed.’ The World Health Organization (WHO) and other non-profit organizations will publicly share the strains of concern – and then vaccine producers design, manufacture, and distribute the vaccine in time for the flu season.” It’s not unusual in influenza for the strain selected to ultimately not end up being the one circulating with high prevalence during the winter seasons.”Įxplaining one of the biggest disadvantages of traditional egg-based processes, Joseph Payne, CEO, President, and Co-Founder of Arcturus Therapeutics, which is developing mRNA-based vaccines and therapeutics, points to another element of speed. There is a limited window of time from when the yearly influenza strain is detected, which puts pressure on the process. In terms of development, strain selection is a challenge too. “Egg-based processes are slow and require huge facility footprints, which are also becoming unfavorable as more attention is paid to sustainability. According to Piergiuseppe Nestola, Manager of Process Technology Consultants at Sartorius, the influenza vaccine has always been a key vaccine to optimize and improve, particularly in terms of the manufacturing process. One example of innovation in this field is Flublok – a protein-based vaccine made in insect cells. That said, some companies were starting to evaluate other options, including a shift from egg-based to cell-culture based vaccines. 
It was well accepted that more modern processes could be used for influenza vaccine production, but innovation in this space has been slow for a variety of reasons, including the old adage: “If it ain’t broke, don’t fix it (especially if the latter needs investment).” Prior to COVID-19, vaccines were not a hot topic, nor were they frequently seen as a lucrative business prospect. The virus is grown in the chorio-allantoic membrane which surrounds the embryo of a fertilized egg in a well-established process that has been in use for decades. As many of you will know, the vast majority of influenza vaccines are traditionally made in chicken eggs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
